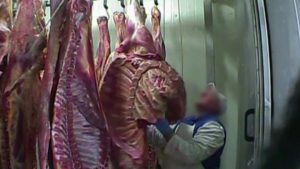
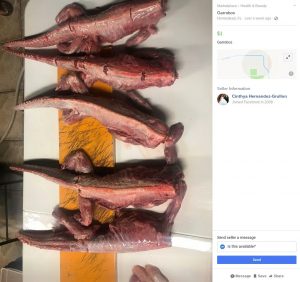
Several postings popped up this week in areas like Miami, Doral and Homestead advertising the animals’ meat for sale.
 Nicole Darrah of The Sun writes some of the animals were seen skinned and beheaded while others were seemingly frozen — indicating they were for sale long before a cold snap hit the Miami area.
Nicole Darrah of The Sun writes some of the animals were seen skinned and beheaded while others were seemingly frozen — indicating they were for sale long before a cold snap hit the Miami area.
One ad, as noted by the Miami Herald, was posted on Facebook’s Marketplace with “garrobo,” another word for lizard used in Latin American countries, being sold for $1.
Iguanas have been spotted all over Florida grounds, parking lots and pools after low temperatures in the 30s and 40s F hit the Sunshine State.
The National Weather Service on Tuesday warned locals about “iguanas falling from trees” — not something they typically forecast.
Low temperatures stun — they don’t necessarily kill — iguanas, which are an invasive species in Florida.
If the animals are hanging out in trees when they’re stunned, they lose their grip and fall to the ground below.
When temperatures warm up, most of the iguanas that were stunned are expected to wake up.
Because iguanas are cold-blooded, the cold weather causes their metabolism to slow down. The reptiles become lethargic as temperatures drop from the warm weather they’re used to.
Iguanas, or “chicken of the trees,” are considered an “economical source of protein” by the University of Florida’s Institute of Food and Agricultural Sciences.